AstraZeneca Initiates Global Recall of COVID-19 Vaccine
In a significant move that reverberates across the global vaccination landscape, pharmaceutical giant AstraZeneca has announced a worldwide recall of its COVID-19 vaccine
HOT: » Which party would you vote for (if you could) in the upcoming snap vote in Bulgaria on April 19?

About 1,500 of the initial volunteers in a late-stage clinical trial of the Oxford/AstraZeneca COVID-19 vaccine were given the wrong dose, but weren’t informed that a mistake had been made after the blunder was discovered, documents obtained by Reuters show.
Instead, the dosing mishap was presented to the trial participants in a letter dated June 8 as an opportunity for University of Oxford researchers to learn how well the vaccine works at different doses. The letter was signed by the trial’s chief investigator, Oxford professor Andrew J. Pollard, and sent to the trial subjects.
On Dec. 24, participants were given about a half dose due to a measuring mistake by Oxford researchers. The Pollard letter didn’t acknowledge any error. Nor did it disclose that researchers had reported the issue to British medical regulators, who then told Oxford to add another test group to receive the full dose, in line with the trial’s original plan.
There is no suggestion there was any risk to the health of trial participants.
Much is riding on the British-developed vaccine, which is being rolled out across the UK and has been touted as a low-cost weapon against the pandemic. The jab has come under scrutiny because of the dosing error in the Oxford trial and a paucity of data about its efficacy in older people who are most vulnerable to the virus.
Reuters shared the letter – which it obtained from the university through a Freedom of Information request – with three different experts in medical ethics. The specialists all said it indicates the researchers may not have been transparent with trial participants. Volunteers in clinical trials are supposed to be kept fully informed about any changes.
The spokesman’s suggestion that no error was made is contradicted by documents produced last year by Oxford and its vaccine partner, drugs giant AstraZeneca PLC. In December, Reuters reported that a “Global Statistical Analysis Plan” by Oxford/AstraZeneca, dated Nov. 17 and later published in the scientific journal The Lancet, called the dosing discrepancy “a potency miscalculation.”
The Oxford/AstraZeneca vaccine recently has received authorization for use in a growing number of countries, including the United Kingdom, the European Union and India. The UK became the first country to approve it, and began rolling out the vaccine on January 4.
But questions surrounding the clinical trials continue to plague the vaccine. Last week, Germany’s vaccine committee recommended it should only be given to people aged under 65, while the EU, which authorized it on Friday for people aged 18 and over, lowered its reported efficacy rate from 70.4% to 60%. In both cases, authorities cited a lack of sufficient data from the clinical trials. The EU also has sharply criticized AstraZeneca for cutting back its planned vaccine deliveries to the continent over the next few months. The company has said it is doing its best to boost supplies.
The mistaken half-dose – which prompted the letter in June to trial participants – continues to be a factor in the reported efficacy of the Oxford/AstraZeneca vaccine.
Oxford had reported interim results in November that showed the efficacy rate for trial subjects who mistakenly received a half-dose and a subsequent full-dose booster shot was 90%, and that the rate for those who received two full doses was 62%. Combining data from the two dosing regimens resulted in an efficacy of 70.4%.
In authorizing the vaccine, UK regulator accepted the pooled results, but didn’t approve administering the half-dose/full-dose regimen. “There is not persuasive evidence of a real difference in” vaccine efficacy between the two different dosing regimens, it said.
The letter, signed by chief investigator Pollard, stated that researchers “are not sure what dose of vaccine is most likely to be protective against COVID disease” and explained that doses “are measured using standard scientific test methods.” It said the late-stage trial participants received a dose measured using one method and that another group will receive a dose measured using a different test to match the dose given in another clinical trial of the vaccine./Reuters

The Bulgarian Medical Association has called for an urgent overhaul of healthcare funding, insisting on a 25 percent increase in the prices of clinical pathways and all medical services, alongside a revision of the user fee for hospital stays
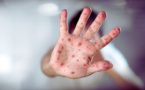
Health authorities report that measles infections in Bulgaria have surpassed 70 confirmed cases, with a total of 73 registered as of April 3
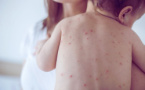
Health authorities have reported a total of 66 confirmed measles cases in Bulgaria, with the first infection now recorded in Sofia, according to data published on the Ministry of Health’s epidemic monitoring platform.
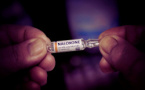
All emergency medical centers across Bulgaria have now been equipped with the opioid antidote Naloxone, a development confirmed by the Ministry of Health. The drug is used in cases of overdose and is considered critical for saving lives in emergencies inv
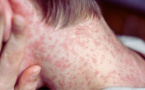
A total of 43 measles infections have been recorded across three regions in Bulgaria, according to data released by the Ministry of Health on March 30
Last year, Bulgaria recorded over 17,000 abortions, according to data compiled by the National Center for Public Health and Analysis

Aniventure Comic Con Returns to Bulgaria with Star Guest Christopher Judge!

Global Fuel Shock: Oil Jumps Over 40% Since Iran War Began